
- Home
- Product category
- Protective
- Protective Face Mask
- KN95 Face Mask
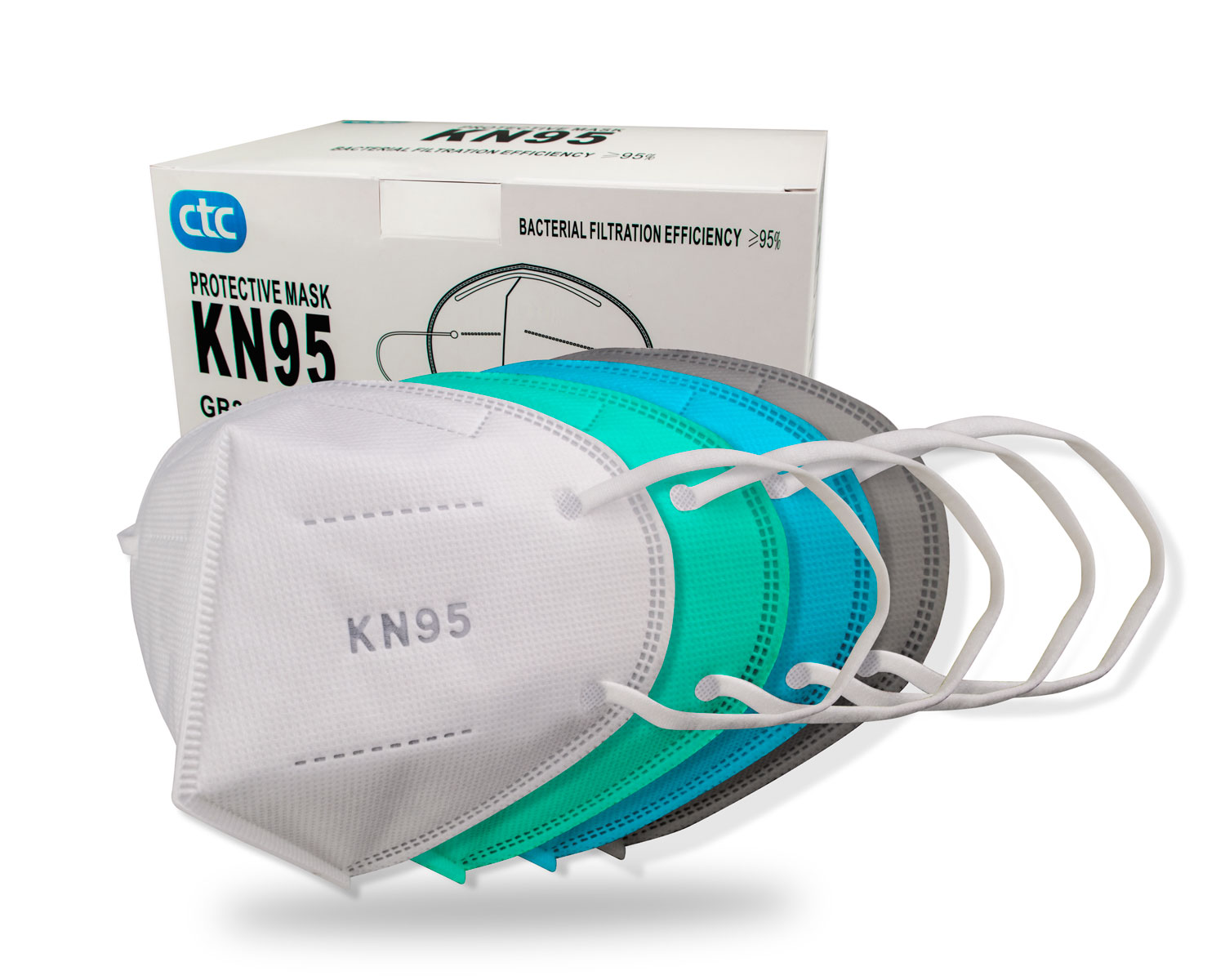
- Wholesale Colored KN95 Non-medical Protective Face Mask



Wholesale Colored KN95 Non-medical Protective Face Mask
description:- Model:CTPL-0020
- Product feature: 5 layer protection, double layer melt-blown fabric
- Packing Method: 2 pcs/bag, 50 pcs/box, 32 boxes/carton
- Small Box Size: 20*13*11cm
- Carton Size: 43.5*41.5*45.5cm
- Single Box Weight: 0.332kg
- Single Carton Weight: 11.9kg
- Certificate:FDA /GB2626-2019
- Expiration date: 2 years
- Storage condition: storage temperature is -30℃~+70℃ and the relative humidity is lower than 80%
- Face mask with filter. Filters at least 95% of airborne particles (0.3 micron) particles.
- Thin & light multi-layer design reducing respiratory resistance.
- Substantial breathing area allows for sufficient airflow circulation without touching lips.
- High filtering ability, extremely low resistance, easy to breathe.
- Flexible & foldable design for extra comfort and easy storage.
- Glue-free and odorless face mask.
- CE Approved KN95 protective mask. Our KN95 face masks have been manufactured to EEA standards, and are used by hospitals, medical facilities and health professionals.
- Soft and flexible nose bridge can be adjusted to the face for comfort and perfect fit.
- Two strap ear-loop design stretches to fit all sizes providing a secure seal.
- Highly compatible with other protective equipment like protective glasses and ear plugs.
- Helps prevent airborne diseases and infections.
1. KN95 – stands for the regulatory standard for filtering facepiece respirators that are certified in China.
2. Regulated by the Chinese government under regulations GB2626-2016, GB2626-2009.
3. 95% protection against all particles that are greater than 0.3 µm in diameter (bacteria, viruses, pollution particles, fine particles, dust, smog, pollen, etc.)
4. 3D foldable design, earloops, nose bridge/nosepiece, and provide excellent fitting and sealing.
5. 5-layer protection, skin-friendly (don’t cause rash and skin irritation).
6. KN95 facemasks are N95 equivalent and meet the guidance from the World Health Organization (WHO).
Product Details





- Before and after touching or adjusting of the respiratory mask, you should clean your hands with soap and water, o ran alcohol-based sanitizer with at least 60% alcohol.
- Avoid touching the inside of the mask.
- When touching and adjusting the mask, make sure to use a pair of clean (non-sterile) gloves.
- Always visually inspect the respiratory mask to see whether its composition and fitting have been compromised.
- Always make sure to check whether the mask components like nose bridge, straps, and nose foam material did not degrade (which can compromise the mask efficiency, quality of the fit, and seal).
- Always make sure to discard a used respiratory mask properly and never leave it exposed, where other people can touch it or use it.
- This product is prohibited to be used with a damaged package;
- Do not use in atmospheres containing less than 19.5% oxygen, as this respirator does not supply oxygen; Not for use in oil mist atmospheres;
- If the product becomes damaged, soiled, or breathing becomes difficult, leave the contaminated area immediately and replace the product;
- This product is one-time use only and can not be washed;
- This product should be stored in a clean, dry, and ventilated environment with relative humidity less than 80% and without harmful gas.
Tips on test fake KN95 mask
1. Flammability – if a mask is KN95 certified, when exposed to a flame it should melt but not ignite. Counterfeit KN95 masks are made from cheap materials and will ignite when in contact with a flame.
2. Permeability – if a mask is KN95 certified, it should limit the airflow going in and out of the mask. You can test this by trying to blow out a candle or a flame of a lighter by blowing through the mask. With a KN95 rated mask, you shouldn’t be able to blow out the flame no matter how hard you blow.
3. Liquid resistance – if a mask is KN95 certified, it will have a waterproof layer that will prevent the passage of fluid. You can test this by pouring some water into the mask. A certified KN95 mask will contain all the water with no leakage, while a counterfeit mask will allow the water to flow through the mask.
We're China factory with ISO, specialized in the production of 5 layers of protective KN95 masks and disposable masks, children masks, protective masks.
It is equipped with 14 facemask production all-in-one machines and related supporting sealing machines and other production equipment. It is equipped with inspection equipment and personnel matching the technical requirements of the products.
1. Nine production lines of KN95 protective masks, with a daily capacity of 500,000.
2. There are 5 production lines of planar masks, with a daily capacity of 800,000.
If you need more information of our company profile and production video, please check, on this link on website.
Check prior to use--The respirator must be selected properly for intended application. An individual risk assessment must be evaluated. Check the respirator that it is undamaged with no visible.
1. Open the mask and check the front and back of the mask.
2. The one with the cap is upward and the ear band is hanging one the ear.
3. The shape of pressing cap is consistent with the nose curve.
4. Perform fit check place hands over the mask and exhale if air leaks, Adjust to the right position.

ISO 9001: 2015 Certified: It sets a global standard for Quality Management Systems and ensures their efficiency for products, services, and international supply chains. It embodies the best practices of Quality Management and Quality certification.
The CNAS accreditation scopes for competence in testing masks, gloves, medical protective clothing and other personal protective equipment for the use of Epidemic prevention, cover both Chinese standards and some foreign standards such as those of EU and USA.
FDA Registered Facility: FDA's Center for Devices and Radiological Health (CDRH) is responsible for regulating firms who manufacture, repackage, relabel, and/or import medical devices sold in the United States. Our facility underwent FDA inspections to assure compliance with the FDA requirements before it was listed as a registered facility.
CONTACT US

Connexions Technology (Dongguan) Ltd.
We are always providing our customers with reliable products and considerate services.
If you would like to keep touch with us directly, please go to contact us






